Self-organizing heart organoids created at the Austrian Academy of Sciences’ Institute of Molecular Biotechnology (IMBA) are also efficient injury and in vitro congenital disease models. These “cardioids” have the potential to transform studies into cardiovascular diseases and heart malformations. The findings were published in the journal Cell.
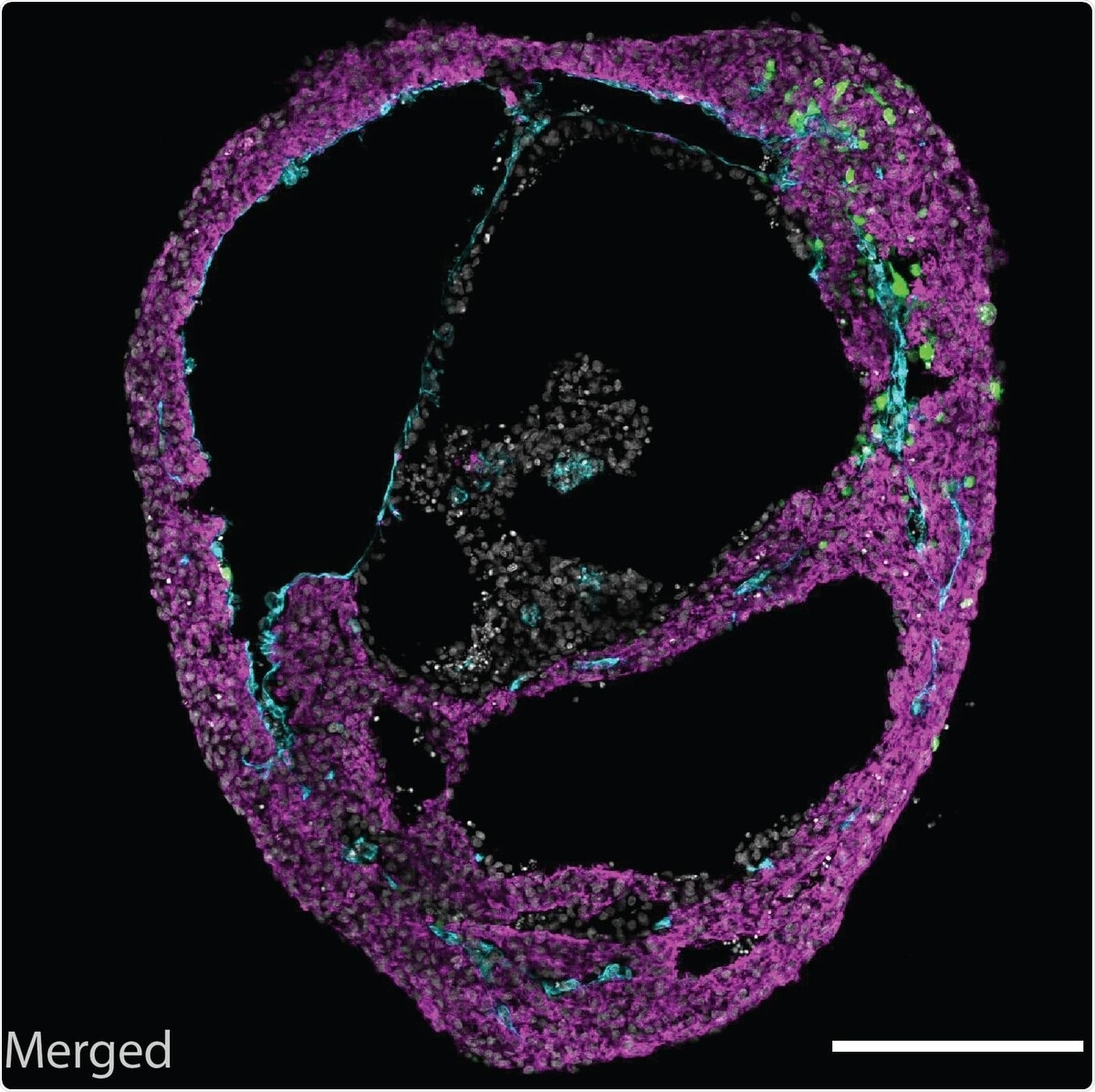
A self-organizing cardioid, containing all three cardiac lineages. Image Credit: Mendjan/Institute of Molecular Biotechnology of The Austrian Academy of Sciences.
Cardiovascular disorders kill almost 18 million people per year, making them the leading cause of death worldwide. Furthermore, the heart is the most common birth defect in infants. Human anatomical models of the heart are still a significant bottleneck in identifying human heart malformations and improving regenerative therapies.
Sasha Mendjan’s research team at IMBA created cardioids, which are human self-organizing cardiac organoids that mimic the lineage architecture of a heart chamber.
Cardioids are a major milestone. Our guiding principle is that for an in vitro tissue to be fully physiological, it also needs to undergo organogenesis. We were able to achieve this, using the developmental principles of self-organization – which makes it such an exciting discovery.”
Sasha Mendjan, Institute of Molecular Biotechnology, Austrian Academy of Sciences
A heart chamber arises from the mesoderm germ layer during growth. The researchers were able to create in vivo-like mesodermal signaling conditions that directed pluripotent stem cells.
Amazingly, this led to self-organization of a heart chamber-like structure that was beating. For the first time, we could observe something like this in a dish. It is a simple, robust and scalable model, and does not require addition of exogenous extracellular matrix like many other organoid models.”
Sasha Mendjan, Institute of Molecular Biotechnology, Austrian Academy of Sciences
A functional heart, in addition to a beating myocardial membrane, has an inner endothelial lining that leads to heart vasculature and an outer epicardial layer that guides heart regeneration and growth. Cardioids mimic the three-layered structure, forming a heart chamber-like structure.
The researchers discovered how transcription factors and signaling influence cardioid chamber development. For example, by destroying a transcription factor related to this defect, the team was able to phenocopy the severe chamber cavity loss seen in children with Hypoplastic Left Heart Syndrome in cardioids.
Cryoinjury (injury by freezing), a procedure that mimics myocardial infarction, was also tested on cardioids. The researchers discovered, for the first time in a dish, that this injury causes an in vivo-like aggregation of extracellular matrix proteins in cardioids, which is an early marker of both fibrotic heart disease and regeneration.
Over the last decade, the field of self-organizing organoids has transformed biomedical science. The heart, however, was the last large inner organ to lack such a physiological model capable of replicating injury response and developmental processes.
Cardioids bear incredible potential to unravel human congenital heart defects. As the system is physiological and scalable, this opens up huge possibilities for drug discovery and regenerative medicine.”
Sasha Mendjan, Institute of Molecular Biotechnology, Austrian Academy of Sciences
A beating cardioid. Video Credit: Mendjan/Institute of Molecular Biotechnology of The Austrian Academy of Sciences.
Source:
Journal reference:
Hofbauer, P., et al. (2021) Cardioids reveal self-organizing principles of human cardiogenesis. Cell. doi.org/10.1016/j.cell.2021.04.034.