Researchers at WEHI have uncovered a never-before-seen mechanism that human bodies utilize to control sugar. These discoveries redefine basic biological principles and pave the way for new scientific discoveries.
 Image credit: max.ku/Shutterstock.com
Image credit: max.ku/Shutterstock.com
The study, published in Nature, shows for the first time that glycogen, the storage form of glucose in the body, can be directly regulated by ubiquitin, a protein traditionally associated with tagging damaged proteins for recycling.
This discovery introduces an entirely new layer of control over sugar metabolism and may have implications for a wide range of conditions, including diabetes, cardiovascular disease, and rare inherited metabolic disorders.
When excess sugar is consumed, the body converts it into glycogen, which is primarily stored in the liver and muscles. For decades, scientists believed glycogen metabolism followed a well-established and fully understood biochemical pathway.
David Komander, co-lead author of the study, stated that the team’s findings add a new chapter to a field previously thought to be fully understood.
It’s quite likely biology books will need to be amended as a result of our findings. We’ve uncovered a second pathway where glycogen can be directly regulated – likely on demand. This is an exciting breakthrough for people living with diseases caused by excessive glycogen.
David Komander, Head, Ubiquitin Signalling Division, The Walter and Eliza Hall Institute of Medical Research
A collection of uncommon hereditary conditions known as Glycogen Storage Diseases (GSD) arises when the body is unable to adequately produce or break down glycogen. They frequently do not have any options for treatment.
More prevalent diseases like diabetes, obesity, liver disease, and heart disease are also associated with excessive glycogen.
Glycogen buildup is what causes these disorders. There are currently no treatments that target the glycogen molecule directly.
Exciting new drugs – such as Ozempic – are transforming how we manage blood sugar, indirectly via hormonal regulation. Without being able to regulate glycogen itself, it is hard to combat its accumulation – the root cause of many diseases. That’s why our study is exciting. We’ve found a way to go straight to the source.
David Komander, Head, Ubiquitin Signalling Division, The Walter and Eliza Hall Institute of Medical Research
The Hidden Hero
Central to the discovery is ubiquitin, a small regulatory protein known for marking damaged or unwanted proteins for degradation.
The study demonstrates that ubiquitin can also attach to glycogen, even though glycogen is a carbohydrate, not a protein. This overturns decades of assumptions about the specificity of ubiquitin signalling.
Glycogen is a sugar rather than a protein. However, this groundbreaking discovery shows that ubiquitin may also bind to sugars in human cells and animal models.
“Ubiquitin is really an unsung hero that has been quietly working in the background all this time, keeping us alive,” Dr. Cobbold said.
The breakthrough was made possible by a novel analytical technique known as NoPro-clipping, developed over several years by the research team. Researchers may now examine ubiquitin in previously unheard-of depth and use mass spectrometry to identify non-protein ubiquitination processes for the first time.
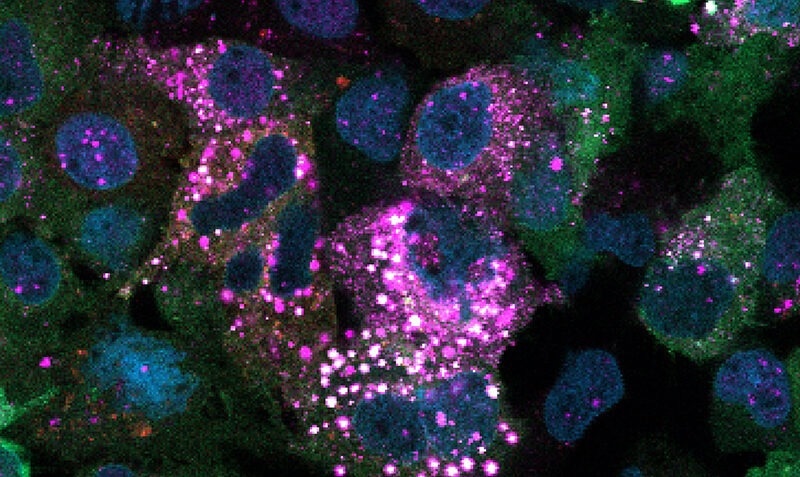 This image shows human liver cells treated with a drug that induces glycogen ubiquitination. The bright white speckles appear where glycogen and ubiquitin overlap, indicating that glycogen has been tagged with ubiquitin inside the cells. Image Credit: The Walter and Eliza Hall Institute of Medical Research.
This image shows human liver cells treated with a drug that induces glycogen ubiquitination. The bright white speckles appear where glycogen and ubiquitin overlap, indicating that glycogen has been tagged with ubiquitin inside the cells. Image Credit: The Walter and Eliza Hall Institute of Medical Research.
Without our tools and method, this remarkable process would have remained invisible. That’s the beauty of NoPro-clipping – it’s allowing us to study a canvas of molecules the ubiquitin field has overlooked all this time.
Dr. Simon Cobbold, The Walter and Eliza Hall Institute of Medical Research
“Not only can we use it to detect ubiquitinated glycogen – we can also uncover ubiquitinated metabolites like glycerol and spermine, which we’ve discovered for the first time in all our cells,” he says.
Jochem notes, “Our discovery is rewriting the fundamental rules of biology and ubiquitin signalling. And I’m sure we’ve only hit the tip of the iceberg.”
Fast Results – How the Study Was Conducted
In a key aspect of the study, the researchers used NoPro-clipping to visualize how ubiquitin attaches to glycogen within the livers of mice under fed and fasting conditions. They observed that during fasting, when energy is required, glycogen levels decrease.
Additionally, the presence of ubiquitin “tags” increased as glycogen levels declined, indicating that sugar ubiquitination plays a role in regulating glycogen breakdown.
These findings reveal ubiquitin as an unexpected component of glycogen metabolism, introducing a new layer to a well-established textbook biochemical process.
Notably, the dynamic changes in sugar-associated ubiquitin tags indicate an important regulatory role in controlling when and how glycogen is released.
The researchers also demonstrated that increasing glycogen ubiquitination led to a reduction of glycogen levels in cells.
If these findings can be applied to animals and humans, they could open new approaches for addressing diseases. Early discussions with investors are already in progress.
Source:
Journal reference:
Jochem, M., et al. (2026). Ubiquitination of glycogen and metabolites in cells and tissues. Nature. DOI: 10.1038/s41586-026-10548-x. https://www.nature.com/articles/s41586-026-10548-x.