Signals from the gut have been linked to autoimmune inflammation, but the fundamental cellular processes underlying this remain poorly understood. A recent investigation from Japan reveals a preserved gut-central nervous system pathway by which intestinal signals drive autoimmune neuroinflammation. These results indicate that focusing on T cell activation in the gut could provide a means to reduce neuroinflammation in diseases such as multiple sclerosis.
 Image credit: New Africa/Shutterstock.com
Image credit: New Africa/Shutterstock.com
Multiple sclerosis (MS) is a severe neurological disease resulting from dysregulated immune responses that attack the brain and spinal cord within the central nervous system (CNS). An inability of the immune system to differentiate ‘self’ from ‘non-self’ components results in heightened autoimmune reactions against self-proteins such as myelin, which serves as a protective sheath surrounding neurons.
Several factors affect the onset and progression of multiple sclerosis, including genetic predisposition, environmental influences, and, more recently, the gut microenvironment. Individuals with MS exhibit alterations in their gut microbiota, while these microbes and their metabolites play a key role in shaping persistent autoreactive immune responses.
However, in attempts to characterize this gut–CNS axis, the cellular mechanisms that relay gut-derived signals to the immune system and modulate autoimmune inflammation in the CNS remain poorly understood.
Recent research from Keio University, published in the journal Science Immunology, reveals a crucial mechanistic role for gut immune responses in driving neuroinflammation.
Increasing evidence shows that the gut microbiota influences neurological diseases such as Parkinson's, Alzheimer's, and MS. However, the mechanisms linking gut microbes, intestinal immunity, and brain inflammation remain unclear. We were keen to identify how gut immune responses contribute to neuroinflammatory diseases.
Tomohisa Sujino, Associate Professor, School of Medicine, Keio University
Expanding on their earlier observation that mild intestinal (ileal) inflammation occurs in experimental autoimmune encephalomyelitis (EAE), a mouse model of MS, the authors aimed to determine whether comparable inflammation is present in patients with MS. Using single-cell RNA sequencing on intestinal biopsies, the team found that inflammatory Th17 cells accumulate in the mouse EAE model as well as in the intestines of patients with MS, indicating a conserved gut–CNS axis that may operate in human disease.
In both EAE mice and individuals with multiple sclerosis, intestinal epithelial cells (IECs) showed increased activity in antigen-presenting pathways. Specifically, epithelial cells in the ileum showed elevated expression of major histocompatibility complex class II (MHC II), which presents antigens to CD4+ T cells, and selective deletion of MHC II in IECs decreased the production of pathogenic Th17 cells and disease severity.
IECs typically do not present antigens to immune cells. Therefore, the team conducted co-culture assays to assess IEC antigen-presenting capacity. The results show that IECs can directly present antigens to CD4+ T cells in the gut via MHC II-dependent mechanisms.
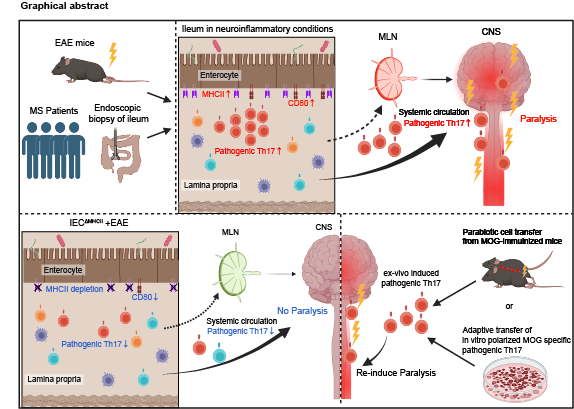
A gut-CNS immunological axis with implications in autoimmune neuroinflammation. In this study, patients with multiple sclerosis (MS) and the experimental autoimmune encephalomyelitis (EAE) mouse model of MS, exhibited an accumulation of Th17 cells in the small intestine. Intestinal epithelial cells upregulate MHC II in response to neuroinflammatory signals, enabling direct antigen presentation to CD4+ T cells and generation of pathogenic Th17 cells that home to the central nervous system (CNS). Created in BioRender. Suzuki, S. (2026). Image Credit: Associate Professor Tomohisa Sujino from Keio University, Japan
Notably, in these assays, IECs promoted Th17 polarization of activated CD4+ T cells. It became clear that the gut acts as a critical site for immune activation of pathogenic CD4+ T cells that differentiate into pro-inflammatory Th17 cells.
To determine whether Th17 cells directly contribute to the pool of autoreactive cells in the CNS, transgenic mice expressing the Kaede protein were used; this protein undergoes photoconversion from green to red fluorescence upon exposure to violet light. This model enabled precise tracking of pathogenic Th17 cells generated in the intestinal lamina propria that subsequently migrate to the spinal cord and drive neuroinflammation.
This study highlights a pivotal role for MHC II expressed by IECs in the expansion of pathogenic Th17 cells that later migrate to the CNS during EAE, establishing a mechanistic connection between gut immune responses and autoimmune neuroinflammatory disorders. This study shows that although systemic circulation permits T cell exchange across immune tissues, epithelial–immune interactions within the gut mucosal environment can fundamentally influence effector T cell responses in the brain.
While current therapies for MS often target B cells, our study highlights the gut as an important therapeutic site. Modulating intestinal microbiota or antigen-presenting activity of IECs represents new approaches to treating autoimmune neurological diseases.
Shohei Suzuki, Assistant Professor, Division of Gastroenterology and Hepatology, Keio University
Source:
Journal reference:
Suzuki, S., et al. (2026) Intestinal epithelial MHC class II induces encephalitogenic CD4 T cells and initiates central nervous system autoimmunity. Science Immunology. DOI: 10.1126/sciimmunol.aec1627. https://www.science.org/doi/10.1126/sciimmunol.aec1627.