From genotoxic bacteria to immune-shaping metabolites, the review reveals how microbes may influence tumor growth, treatment resistance, and the next generation of precision cancer therapies.

Review: The microbiome: regulating anti-tumor immunity. Image Credit: Kateryna Kon / Shutterstock
A recent review published in the journal Frontiers in Immunology highlights the microbiome as a potentially important regulator of cancer progression and therapeutic response. Far from being passive bystanders, microbes shape tumor immunity by influencing inflammation, producing genotoxic compounds, and modulating immune activity within the tumor microenvironment.
Emerging strategies, from targeting microbial toxins to reshaping gut communities, suggest that precision microbiome interventions may help enhance immunotherapy outcomes, address some forms of treatment resistance, and open new avenues for biomarker discovery in cancer care.
Together, these insights suggest that the microbes humans coexist with may play a decisive role in shaping responses to cancer therapy, with direct implications for patient outcomes.
Microbiome, Tumor Immunity, and Cancer Therapy
Microorganisms living in the body support essential functions, including nutrient absorption, metabolic processes, detoxification, and immune defense. Disruptions in these microbial communities, along with their metabolites, are increasingly linked to altered immune activity, influencing both therapeutic response and treatment-related toxicity in cancer.
In parallel, advances in tumor immunology unravel intricate signaling networks and microenvironmental dynamics that shape immunotherapy outcomes.
Clarifying the relationship between microbial balance, immune function, and tumor biology may help refine prognostic tools and reveal new therapeutic targets across diverse cancer types.
In this review, researchers explore the microbiome as a regulator of anti-tumor immunity and highlight emerging strategies to target microbial communities and improve cancer outcomes.
Microbial Mechanisms in Tumor Progression
The microbiome influences tumor immunity via multiple, tightly linked mechanisms that operate at both systemic and local levels. Certain microbes directly damage host deoxyribonucleic acid (DNA) by producing genotoxins, increasing genomic instability and mutation rates.
For example, Escherichia coli strains carrying the polyketide synthase (pks) island generate colibactin, which induces DNA double-strand breaks, while Campylobacter jejuni produces cytolethal distending toxin with similar effects.
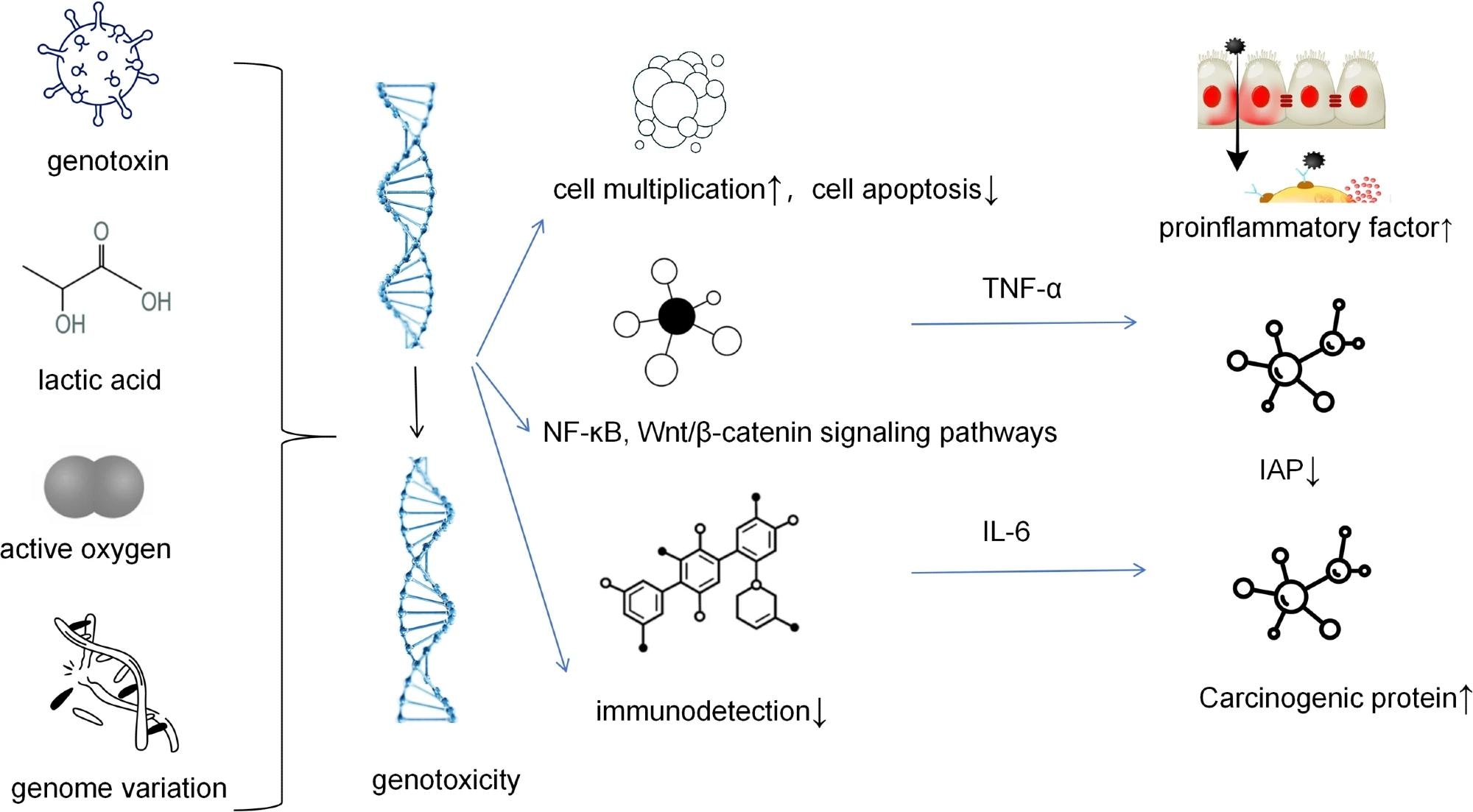
IAP, inhibitor of apoptosis protein. Microorganisms (such as certain strains of Escherichia coli) produce gene toxins such as colibactin, which can directly cause DNA double-strand breaks. This leads to increased genomic instability and is the “initial driving force” for tumor formation. Microorganisms (and cancer cells themselves) metabolize to produce large amounts of lactic acid, creating an acidic microenvironment. This acidic environment not only directly promotes cell proliferation but also inhibits the function of immune cells and provides conditions for epithelial-mesenchymal transition. Microbial infection or metabolic activities induce cells to produce a large amount of reactive oxygen species, which act as a key second messenger, activating signaling pathways such as NF-κB. Activated NF-κB enters the cell nucleus and initiates the transcription of pro-inflammatory factors and anti-apoptotic proteins, jointly creating a microenvironment that promotes cell survival, proliferation, and immune evasion.
Other bacteria, such as Fusobacterium nucleatum, promote tumorigenesis by activating oncogenic pathways such as the wingless-related integration site/β-catenin (Wnt/β-catenin) signaling. They also trigger chronic inflammation through the nuclear factor kappa B (NF-κB) and mitogen-activated protein kinase (MAPK) cascades.
Beyond direct damage, microbes actively shape immune responses within the tumor microenvironment. Lipopolysaccharides (LPS), key components of the outer membrane of Gram-negative bacteria, can activate pattern-recognition receptors (PRRs) on immune cells. This signaling enhances antigen presentation and promotes cytotoxic cluster of differentiation 8-expressing (CD8+) T-cell responses.
Specific taxa further modulate immune infiltration; for instance, members of the Ruminococcaceae group can boost dendritic cell function and improve T-cell-mediated anti-tumor activity. Conversely, dysbiosis can foster immunosuppression by expanding regulatory T cells, myeloid-derived suppressor cells (MDSCs), and M2-like macrophages, all of which dampen effective immune responses.
Microbial metabolites add another layer of regulation. While some compounds enhance immune surveillance, others promote immune escape, angiogenesis, and metastasis under tumor-driven selective pressure.
Persistent microbial signals can also maintain inhibitory immune pathways, including cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein-1 (PD-1), ultimately contributing to T-cell exhaustion.
Together, these findings underscore that the microbiome is not a passive bystander but an active regulator of tumor immunity, capable of both constraining and promoting cancer progression depending on its composition and functional balance.
Microbiome-Targeted Cancer Treatment Strategies
Efforts to therapeutically target the microbiome are rapidly gaining momentum as evidence links tumor-associated microbes to treatment response, resistance, and disease progression. Researchers are now exploring strategies that selectively modulate microbial activity without disrupting overall ecological balance.
For instance, inhibiting genotoxins such as colibactin produced by E. coli represents a promising approach to limit DNA damage while preserving beneficial microbes.
Microbial metabolites also offer therapeutic leverage. Short-chain fatty acids, including butyrate and propionate, can enhance CD8+ T-cell function and promote pro-inflammatory M1 macrophage polarization, thereby strengthening anti-tumor immunity.
However, not all metabolites are beneficial. Compounds such as trimethylamine N-oxide (TMAO) and secondary bile acids exhibit context-dependent effects, either enhancing immune responses or promoting tumor growth, underscoring the need for precision targeting.
Microbiome-based interventions are beginning to move through clinical and translational studies. Fecal microbiota transplantation (FMT) has shown potential to help restore responsiveness to immune checkpoint inhibitors in some melanoma patients.
Similarly, specific bacterial taxa, such as Akkermansia muciniphila and Faecalibacterium prausnitzii, have been associated in some studies with stronger responses to immune checkpoint blockade, although these associations are not yet validated as stand-alone predictive biomarkers.
Emerging approaches include engineered bacteria, live biotherapeutic products, and prebiotic or dietary interventions designed to reshape microbial communities and enhance the efficacy of immunotherapy.
Microbiome Precision Oncology Challenges and Outlook
The findings position the microbiome as a critical modulator of tumor immunity and treatment response, offering new opportunities to refine cancer therapy. Despite this progress, translating these insights into clinical practice remains challenging due to inter-individual variability and the complexity of microbial ecosystems.
The authors also caution that tumor microbiome research requires rigorous validation, because low-biomass samples, contamination, batch effects, and background microbial signals can complicate interpretation. Many microbiome-cancer links remain associative, and further studies are needed to clarify whether specific microbial communities drive tumor behavior or reflect changes in the tumor microenvironment.
Future strategies will likely integrate microbial profiling with tumor genomics, metabolism, and host immunity to enable more precise, personalized interventions.
Approaches such as dietary modulation, targeted probiotics, and fecal microbiota transplantation show promise, but require validation in well-designed clinical trials to ensure safety and long-term efficacy.
Download your PDF copy by clicking here.