Huntington’s disease is a progressive neurological disorder that impairs movement, cognition, and behaviour over time. It is driven by a toxic protein that accumulates within brain cells, ultimately leading to their dysfunction and death. Researchers have long recognised that this protein can pass between cells, contributing to disease progression, but the mechanisms behind this spread, and how to prevent it, have not yet been fully understood.
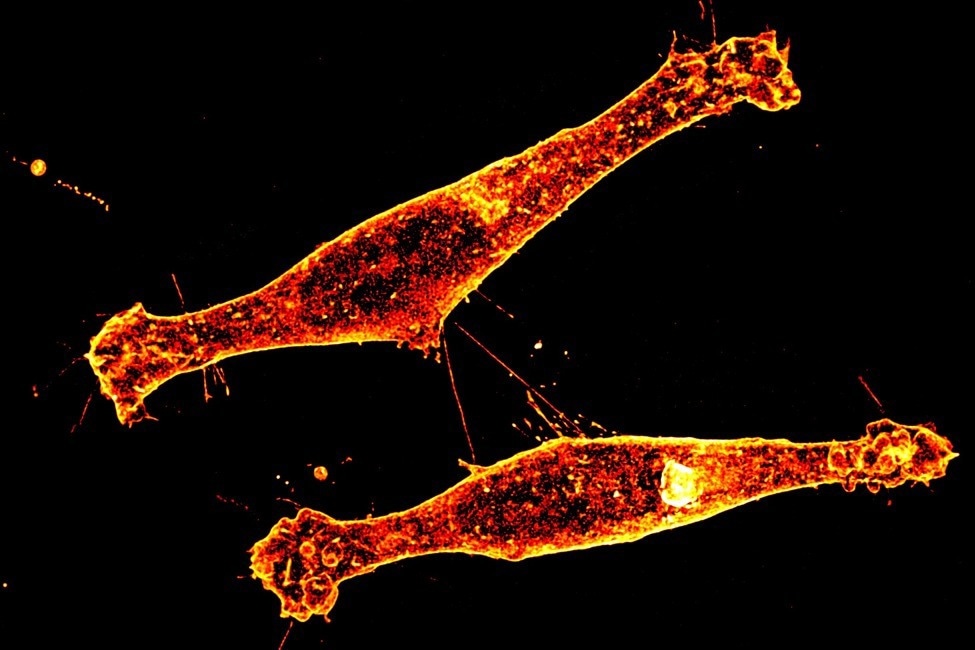 Tunneling nanotubes form connections between brain cells that express Rhes, a protein linked to Huntington’s disease. Image Credit: Gisele Galoustian, Florida Atlantic University
Tunneling nanotubes form connections between brain cells that express Rhes, a protein linked to Huntington’s disease. Image Credit: Gisele Galoustian, Florida Atlantic University
Researchers from Florida Atlantic University and collaborators have made a significant discovery: a previously unidentified cellular mechanism that enables brain cells to transfer toxic materials directly to their neighbors via tiny, tube-like structures. Crucially, the study that was published in Science Advances demonstrates that blocking this pathway significantly slows the spread of the protein that causes the disease in the brain.
These tiny structures, known as "tunneling nanotubes," serve as direct channels between cells. Unlike chemical signals that travel over space, nanotubes enable cells to transfer proteins and other materials by hand-delivery. While this type of sharing can help healthy cells adapt to stress or injury, it can also be detrimental if it transmits harmful proteins, such as the mutant huntingtin protein that causes Huntington’s disease.
The research has uncovered that a protein named Rhes, which is already recognized for its significant involvement in Huntington’s disease, collaborates with an unexpected partner: a bicarbonate transporter known as SLC4A7, a protein primarily recognized for its role in helping cells manage their internal acidity. Together, these two proteins contribute to the formation of tunneling nanotubes, establishing pathways that enable the transfer of toxic huntingtin protein between neurons.
This work fundamentally changes how we think about disease progression in Huntington’s. We’ve known that neurons somehow pass toxic proteins to one another, but now we can see the machinery that makes that possible. By identifying SLC4A7 as a key partner of Rhes, we’ve uncovered a new and potentially druggable target to stop that spread at its source.
Srinivasa Subramaniam, PhD, Study Senior Author and Associate Professor, Department of Chemistry and Biochemistry, Charles E. Schmidt College of Science, Florida Atlantic University
Using sophisticated protein-mapping methods, the researchers revealed that Rhes physically interacts with SLC4A7 on the cell membrane. When this relationship occurs, changes occur within the cell that stimulate the formation of nanotubes. When the researchers blocked SLC4A7, either genetically or with drugs, the nanotubes failed to form, and the hazardous huntingtin protein was mainly prevented from spreading.
Significantly, this impact was not limited to isolated cells. In Huntington's disease mouse models, SLC4A7-deficient mice showed a significant decrease in harmful protein transfer across striatal neurons, the brain area most affected by the disease. This implies that interfering with this newly found mechanism might limit the course of Huntington's disease by preventing damage from spreading.
The consequences of this study go well beyond Huntington's disease. Tunneling nanotubes have been linked to various neurodegenerative disorders, including conditions involving tau protein, as well as cancer, where tumor cells share signals, energy, and even drug resistance through similar structures. Since both Rhes and SLC4A7 are involved in key cellular activities, the newly discovered pathway might be a common mechanism underpinning the development of damage in numerous diseases.
This research shines a spotlight on an entirely new way cells communicate in health and disease. By learning how harmful proteins physically move from cell to cell, we gain powerful new leverage points for therapy. The idea that we could slow or even halt disease progression by blocking these microscopic tunnels opens an exciting frontier for treating not only Huntington’s disease, but a wide range of neurological disorders and cancers in the future.
Randy Blakely, PhD, Executive Director, Stiles-Nicholson Brain Institute
As scientists continue to investigate how cells communicate information and how that sharing might go awry, this discovery provides new optimism that disease prevention will one day be as simple as closing the door between cells.
Huntington’s disease is a rare, hereditary brain condition that impacts approximately three to seven individuals per 100,000 globally, affecting both men and women equally. Symptoms typically manifest between the ages of 30 and 50 and progressively worsen, leading to uncontrollable movements, cognitive deterioration, and severe psychiatric issues.
Huntington's disease is a rare, inherited brain disorder that affects three to seven individuals per 100,000 people globally, with men and women affected equally. Symptoms often develop between the ages of 30 and 50 and intensify with time, resulting in uncontrollable movements, cognitive deterioration, and severe mental problems.
Each child of an affected parent faces a 50 % risk of inheriting the disorder. There is currently no cure, and available treatments focus on managing symptoms rather than slowing disease progression. Following the onset of symptoms, individuals typically live for 10 to 20 years, often experiencing a steady decline in function and independence.
Research Video of Tiny Cell 'Tunnels'
The proteins SLC4A7 and Rhes help move the toxic mutant huntingtin protein from one brain cell to another through tiny tube-like connections called tunneling nanotubes. The transfer happens quickly – within minutes – and is shown by the yellow or white arrowheads. Video Credit: Emaad Mirza, Florida Atlantic University
Source:
Journal reference:
Dagar, S., et al. (2026) Membrane-associated Rhes-Slc4a7 complex orchestrates tunneling nanotube formation and mutant Huntingtin spread. Science Advances. DOI: 10.1126/sciadv.aea1226. https://www.science.org/doi/10.1126/sciadv.aea1226