A multinational team of researchers has made a significant advance in the manufacturing of doxorubicin, a crucial chemotherapy drug. The research finds and addresses molecular “bottlenecks” that have hampered the natural manufacturing of this drug for over 50 years.
 Image credit: Beyond This/Shutterstock.com
Image credit: Beyond This/Shutterstock.com
Doxorubicin, a chemotherapy drug, was originally licensed for medical use in the 1970s. It is a key treatment for a variety of cancers, including breast cancer, bladder cancer, lymphomas, and carcinomas, with over one million people receiving it each year. Bacteria naturally generate this vital drug, although inefficiently. As a result, the pharmaceutical industry has depended on costly, multi-step semisynthetic procedures.
We have uncovered several independent factors that limit the formation of doxorubicin. By addressing these bottlenecks, we have harnessed rational strain engineering to pave the way for cost-effective manufacturing that can meet growing global demand.
Keith Yamada, PhD, Study Lead Scientist and Researcher, University of Turku
Researchers Develop New Strain of Bacteria that Enhances Drug Production
The study was the outcome of a major international partnership that included six research laboratories: the University of Turku in Finland, three in the United States, and two in Leiden, the Netherlands.
The teams discovered three key restrictions that limit the yield of doxorubicin manufacturing.
First, the scientists discovered the specific natural "biological power supply", redox partners dubbed Fdx4 and FdR3, that provide the electron flow required to power the drug-producing enzyme.
Second, they discovered that a protein known as DnrV functions as a "molecular sponge" that binds drugs. It sequesters (binds and retains) doxorubicin, so the medication does not inhibit the enzyme's own biosynthetic pathway.
Finally, using X-ray crystallography, the scientists observed the enzyme for the first time, revealing that the drug molecule is located in an unfavorable position within the enzyme, which explains the sluggish reaction rate.
By integrating their findings, the researchers created a novel strain of bacteria that generates 180 % more doxorubicin than existing industry standards.
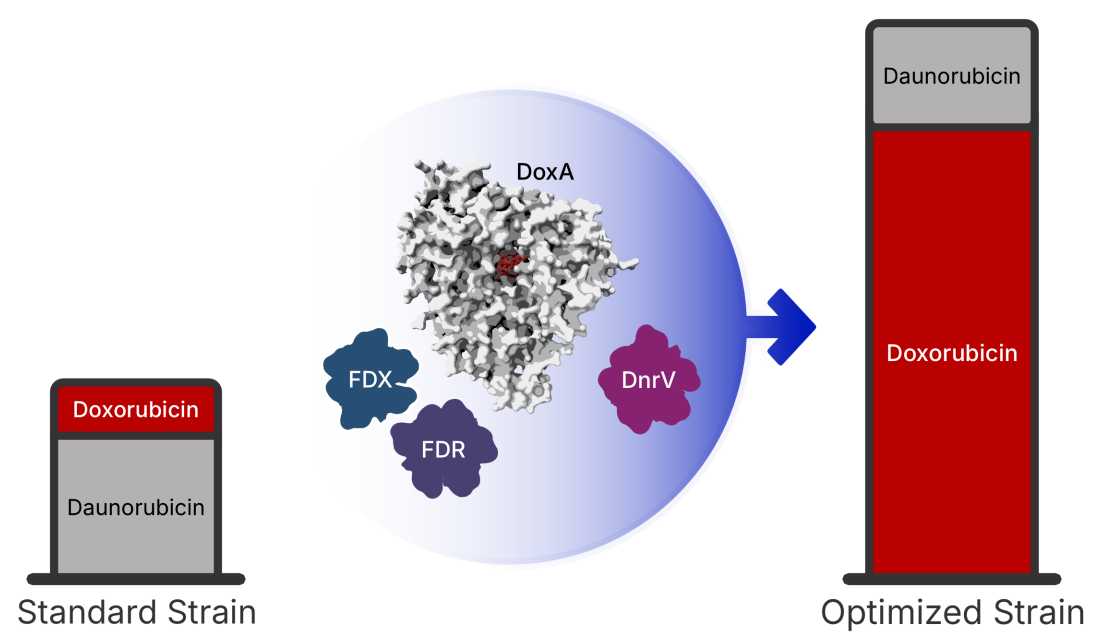 The image shows how the new, optimized strain of bacteria produce an increased amount of doxorubicin. In the center, the primary production machinery (DoxA) is enhanced by a biological 'power supply' (FDX and FDR) and a 'molecular sponge' (DnrV). While the power supply keeps the process running at high speed, the sponge prevents the drug from clogging the system, allowing the engineered cells to produce the medicine with unprecedented purity and a 180 % higher yield than standard industrial methods. Image Credit: Keith Yamada
The image shows how the new, optimized strain of bacteria produce an increased amount of doxorubicin. In the center, the primary production machinery (DoxA) is enhanced by a biological 'power supply' (FDX and FDR) and a 'molecular sponge' (DnrV). While the power supply keeps the process running at high speed, the sponge prevents the drug from clogging the system, allowing the engineered cells to produce the medicine with unprecedented purity and a 180 % higher yield than standard industrial methods. Image Credit: Keith Yamada
Meta-Cells Oy, a spin-off company from the University of Turku, was founded last year to put these results into practice. The company intends to commercialize these cutting-edge technologies for the long-term production of vital antibiotics and anti-cancer drugs. This transition to entirely biosynthetic production offers a cleaner, more consistent supply of life-saving drugs.
Source:
Journal reference:
Koroleva, A. et al. (2026) Metabolic engineering of doxorubicin biosynthesis through P450-redox partner optimization and structural analysis of DoxA. Nature Communications. DOI: 10.1038/s41467-026-69194-6. https://www.nature.com/articles/s41467-026-69194-6.