Hematopoietic stem cells (HSCs), the cells that produce all blood cell types, begin to lose their efficacy as we age, eventually reducing the ability to maintain healthy blood and a robust immune system.
 Image credit: BioFoto/Shutterstock.com
Image credit: BioFoto/Shutterstock.com
HSCs can normally self-renew and produce a balanced mix of blood cells, but over time, they become unable to sustain a strong immune response, produce fewer new cells, and favor some cells over others, such as myeloid cells over lymphoid cells.
This decrease appears to be caused by accumulated cellular damage, altered gene expression, persistent low-level inflammation, and alterations in the bone marrow environment. Nevertheless, it remains unknown exactly how these various pressures interact to impair HSCs.
Investigators from The University of Tokyo, Japan, and St. Jude Children's Research Hospital, USA, focused on the receptor-interacting protein kinase 3 (RIPK3)-mixed lineage kinase like (MLKL) signaling axis, a pathway typically linked to necroptosis, or programmed cell death, in an effort to identify a mechanism explaining how age-related stresses drive HSC functional deterioration. The findings, published in Nature Communications, later emphasized that MLKL plays a non-lethal role.
We discovered an unexpected phenotype in HSCs of MLKL-knockout mice repeatedly treated with 5-fluorouracil, where aging-associated functional changes were markedly attenuated despite no detectable difference in HSC death, prompting us to investigate whether this pathway might induce functional changes beyond cell death.
Dr. Masayuki Yamashita, Assistant Professor, Institute of Medical Science, University of Tokyo
The scientists used a combination of functional assays, stress treatments, and genetic mouse models. They employed RIPK3-deficient, MLKL-deficient, and wild-type mice, as well as specialized reporter mice that could detect MLKL activation via an energy-transfer-based Förster resonance energy transfer (FRET) biosensor.
Mice were subjected to stimuli that resemble aging, such as oncogenic stress, replication stress, and inflammation. The primary method used to evaluate HSC function was bone marrow transplantation, which gauges the stem cells' capacity to repair the blood system.
Complementary analyses included flow cytometry, ex vivo expansion, RNA-seq, assay for transposase-accessible chromatin-seq, high-resolution microscopy, metabolic assays, and mitochondrial analyses, enabling a detailed understanding of how non-lethal MLKL activation impairs HSC function at molecular, cellular, and organelle levels.
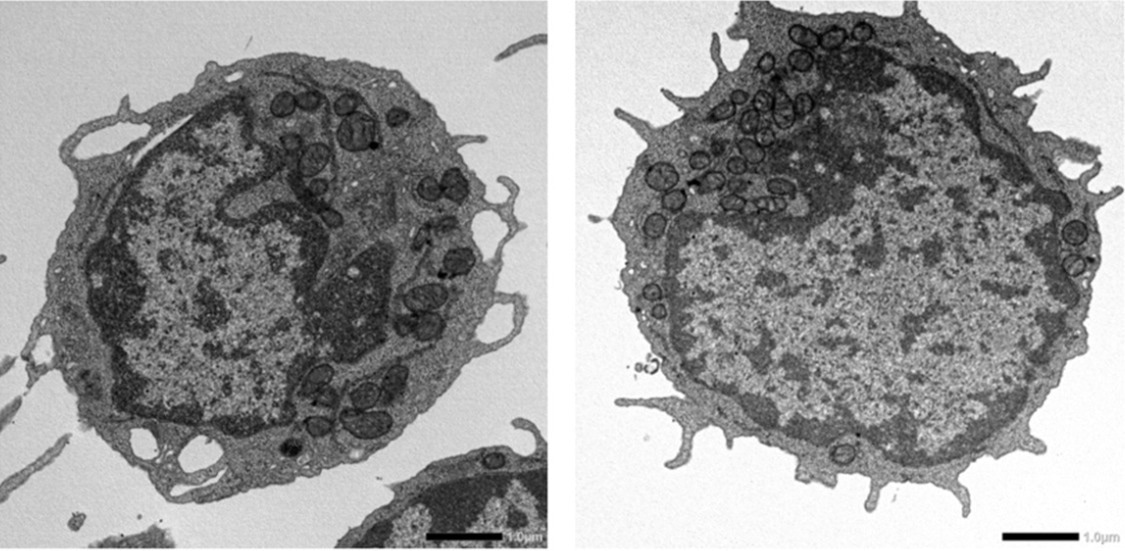 MLKL-Dependent Mitochondrial Changes in Aged Hematopoietic Stem Cells. Transmission electron microscopy images of hematopoietic stem cells from 18 month old wild type (left) and MLKL deficient (right) mice. The images were captured by Dr. Yuta Yamada and Dr. Masayuki Yamashita in collaboration with Dr. Yuji Watanabe and Dr. Hiroshi Sagara at Medical Proteomics Laboratory, The Institute of Medical Science, The University of Tokyo, Japan. Image Credit: Dr. Masayuki Yamashita from The University of Tokyo, Japan
MLKL-Dependent Mitochondrial Changes in Aged Hematopoietic Stem Cells. Transmission electron microscopy images of hematopoietic stem cells from 18 month old wild type (left) and MLKL deficient (right) mice. The images were captured by Dr. Yuta Yamada and Dr. Masayuki Yamashita in collaboration with Dr. Yuji Watanabe and Dr. Hiroshi Sagara at Medical Proteomics Laboratory, The Institute of Medical Science, The University of Tokyo, Japan. Image Credit: Dr. Masayuki Yamashita from The University of Tokyo, Japan
The findings demonstrated MLKL's new, non-necroptotic function in HSC aging. Although MLKL is usually associated with cell death, its activation in HSCs did not result in either decreased cell counts or increased cell death. Rather, stress-induced MLKL activation was short-lived and restricted to mitochondria, where it directly damaged them by lowering membrane potential, altering mitochondrial structure, and limiting energy output. Due to these alterations, HSCs began to show signs of aging, including decreased lymphoid differentiation, decreased self-renewal, and a shift toward myeloid-biased output.
Importantly, MLKL loss or inactivation greatly reduced these abnormalities. Even under stress or in elderly mice, MLKL-deficient HSCs retained their ability to regenerate, generated healthier immune cells, showed less DNA damage, and maintained mitochondrial function.
It is interesting to note that these improvements happened without significant changes in chromatin accessibility or gene expression, indicating that MLKL predominantly causes HSC aging through post-transcriptional and organelle-level mechanisms rather than through transcriptional regulation or inflammation.
These findings have broad implications for understanding aging and potential therapies. By linking diverse stress signals to mitochondrial dysfunction via MLKL, the study identifies a common pathway underlying HSC aging.
In the longer term, this research could lead to therapies that preserve the function of hematopoietic stem cells, ultimately improving recovery and long-term health for patients undergoing chemotherapy, radiation, or transplantation. By revealing how non-lethal activation of cell-death pathways drives stem cell aging, these findings may inspire new classes of mitochondrial-protective or necroptosis-modulating drugs.
Dr. Masayuki Yamashita, Assistant Professor, Institute of Medical Science, University of Tokyo
This research reveals a previously unknown function of MLKL as a non-lethal regulator of stem cell aging. MLKL functions as a stress-responsive factor that damages mitochondria and reduces HSC function, rather than causing cell death. These discoveries not only reinterpret the function of necroptosis-related proteins but also create new opportunities to comprehend and possibly treat hematopoietic system aging.
Source:
Journal reference:
Yamada, Y., et al. (2026). Non-necroptotic MLKL function damages mitochondria and promotes hematopoietic stem cell aging. Nature Communications. DOI: 10.1038/s41467-026-71060-4. https://www.nature.com/articles/s41467-026-71060-4.